Ĭontrolled room temperature implies a certain degree of control over the temperature of the storage conditions in that extremes of hot and cold temperatures are not encountered.Īs a regional director, they're putting the responsibility directly on me for accomplishing it.Ĭontrolled room temperature is defined as 25 C (77F) excursions are permitted between 15 C and 30 C (59F to 86F). STORAGE: Controlled room temperature 20° to 25☌ (68° to 77☏).
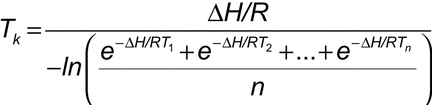
Controlled room temperature, refrigeration, freezing), storage areas should be equipped with temperature recorders or other devices that will indicate when the specific temperature range has not been maintained. When specific temperature storage conditions are required (i.e. Webinar participants can ask about best practices for the MKT calculation in their controlled environments and will become familiar enough with the value to be able to use it to interpret their own data, and explain their decisions to an auditor.All prescription drugs shall be stored at appropriate temperatures and under appropriate conditions in accordance with requirements, if any, in the labeling of such drugs or with requirements in the current edition of an official compendium.17.11(1) Controlled room temperature.Ĭontrolled room temperature means a temperature maintained thermostatically that encompasses the usual and customary work- ing environment 20° to 25° Celsius (68° to 78° F).Ĭontrolled room temperature is defined as 25C (77 F) excursions are permitted between 15C and 30C (59F to 86F).All drug products should be stored in a securely locked area, accessible only to authorized site personnel.Ĭontrolled room temperature is defined as 25 C (77 F) excursions are permitted between 15 C and 30 C (59 F to 86 F).All drug products should be stored in a securely locked area, accessible only to authorized site personnel.Ĭontrolled room temperature is defined as 25☌ (77☏) excursions are permitted between 15☌ and 30☌ (59☏ and 86☏). For example, if we understand why MKT was originally used to select temperatures for long-term stability studies, then we understand why MKT is suitable for evaluating the long-term temperature performance of ambient storage areas. Participants will leave the webinar with an understanding of MKT as a non-linear weighted average that shows the effects of temperature variations over time. Vaisala’s Senior Regulatory Expert Paul Daniel will provide you with 6 clear Do’s and Don’ts for using the Mean Kinetic Temperature calculation, including where it is useful in decision-making for regulated industries, and where it is not. We also discuss the regulatory guidance that describes where and when it is appropriate to use the mean kinetic temperature calculation, including FDA, EMA, and ICH. In this webinar we outline the history of the mean kinetic temperature calculation in pharmaceutical manufacturing and distribution. Also the regulatory guidance that describes where and when to use the mean kinetic temperature calculation: FDA, EMA, and ICH. Kinetic energy Definition & Formula Britannica. 2-Channel and 5-Channel RF Temperature loggers for Validation. where TMKT is the mean kinetic temperature in degrees Kelvin, H is the activation energy in kJmol1. Abstract: Mean kinetic temperature, a thermodynamically calculated average temperature that is used to determine whether or not the controlled room temperature drug storage limit has been maintained to ensure drug stability, first became official in the Ninth Supplement of the United States PharmacopeiaNational Formulary on November 15, 1993. Mean kinetic temperature MKT is a simplified way of expressing the overall effect of temperature fluctuations during storage or.

The history of the mean kinetic temperature calculation in pharmaceutical manufacturing and distribution. The reaction progress can be defined as follows.
